Dernière mise à jour : 16 janvier 2018
Détails
État du projet:
Terminé
Gamme de produits:
Analyse prospective
Sous-type de projet :
Rapport sur une technologie de la santé émergente
Numéro de projet :
EH0060-000
(Document seulement disponible en anglais à l'exception du résumé)
Résumé
- Le glioblastome est le type de cancer du cerveau primitif le plus fréquent chez les adultes.
- Malgré les progrès réalisés dans les traitements, l’espérance de vie des patients atteints de glioblastome est normalement de moins de deux ans.
- Optune est une technologie portable destinée à être utilisée en complément des traitements classiques — résection chirurgicale, radiothérapie et chimiothérapie — chez les adultes lorsqu’un nouveau diagnostic de glioblastome est posé ou en présence d’un glioblastome récurrent. Il a aussi été testé en tant que traitement unique chez les patients adultes atteints d’un glioblastome récurrent.
- Optune fournit des champs électriques alternatifs, ou « champs de traitement de la tumeur » au cerveau afin de perturber la division des cellules cancéreuses.
- Les preuves laissent croire que certains patients ayant reçu un nouveau diagnostic de glioblastome qui sont traités par la technologie Optune pourraient vivre plus longtemps, bien qu’il soit difficile de déterminer avec assurance quels patients sont les plus susceptibles d’en bénéficier.
- Le cout du traitement avec le système Optune est d’environ 21 000 $ US par patient, par mois.
Background
Glioblastoma is also called glioblastoma multiforme (or GBM), Grade IV glioma, or Grade IV astrocytoma.1 In adults, glioblastoma is the most common primary brain cancer (a cancer that originates in the brain).2,3 Glioblastoma is difficult to treat because the cancer cells infiltrate normal brain tissue with tentacle-like extensions that usually cannot be completely removed surgically. Delivery of drug therapies to the tumour is also impeded by the blood-brain barrier (which protects the brain from harmful substances in the blood).4,5 And because glioblastoma cancer cells are highly heterogeneous (that is, when they divide, the new cells are not genetically identical to the parent cells), new tumour growth can contain cancer cells that are resistant to treatment.4,5
With standard treatments, patients with glioblastoma have a median survival of about 15 months from the time they are diagnosed, and only one in four patients is alive at two years.4,6-11 Glioblastoma typically recurs within about seven months of initial treatment.6,10
A wearable device that emits low-intensity, alternating electric fields is a new addition to treatment options for some patients with glioblastoma.
The Technology
The Optune system (Novocure, St. Helier, Jersey Isle, UK), initially called NovoTTF-100A, was developed at the Technion – Israel Institute of Technology. It uses pre-set, low-intensity (one to three volts) alternating electrical fields at an intermediate frequency of 200 kHz to create “tumour-treating fields” (TTFields). These TTFields penetrate cell walls to disrupt rapid cancer cell division and cause cell death,12-14 although the mechanism(s) of action are still not fully understood.15-17 The TTFields treatment should be administered continuously, for at least 18 hours per day.14
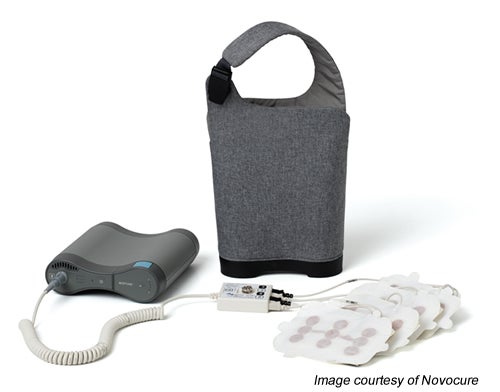
The components of the Optune system include:
- four adhesive patches, called transducer arrays, that are worn on the patient’s head, with each array containing nine, insulated ceramic disks
- a connector cable
- a portable electric field generator
- four rechargeable battery packs (the system uses one battery at a time, each battery providing four to six hours of power)
- a customized backpack.18,19
The Optune system allows the patient to be mobile, using the battery pack and backpack, while receiving treatment, or the system can be plugged into a regular electrical outlet when the patient is seated or asleep.19 It weighs about 1.3 kg, including the battery.18,20
Patients must shave their heads to allow the transducers to properly adhere to their scalp with a thin layer of conductive hydrogel similar to that used for electrocardiogram pads.19,21 The transducer arrays are attached to adhesive bandages to hold them in place and are positioned in pairs on the head left and right, front and back.13,14 Each transducer array is plugged into the connector cable, which is in turn plugged into the battery pack.19 Replacement transducers are provided in sterile packages, and used transducers are returned to Novocure for disposal.1,19 Ideally, the arrays are worn continuously for three to four days; but after exercise, or in hot or humid weather, more frequent exchanges may be needed.22 The scalp must be shaved and cleansed before a new set of arrays can be applied.19,22 Patients can wear a loosely fitting wig, scarf, or hat over the arrays, but the wires will still be visible.23
To help physicians identify the optimal placement location for the arrays on the patient’s head, the manufacturer provides the NovoTAL System software. This software uses data from the patient’s MRI scans to guide placement, and it can also be used to make adjustments to placement, as needed, throughout treatment.6,8
Availability
The Optune system does not currently have a Health Canada Medical Devices Active Licence. However, the system has been used at Canadian hospitals in Ontario, Quebec, Manitoba, and Alberta as part of the multinational EF-14 trial.24
The US FDA first approved the Optune system in April 2011 for use in patients older than 22 years with recurrent glioblastoma, after standard medical therapies have been used.25 In 2015, the FDA expanded the licensing to include the treatment of patients with newly diagnosed glioblastoma, after “maximal debulking surgery and completion of radiation therapy together with concomitant standard of care chemotherapy.”25,26 The Optune system is also commercially available in Europe, Israel, and Japan.26,27
Cost
A 2016 report by the ECRI Institute notes that the cost of TTFields therapy is substantial — approximately US$21,000 per patient each month.28 The cost includes the Optune system, a month’s supply of transducer arrays, and 24-hour technical support.29 The final EF-14 trial analysis showed the median duration of TTFields treatment was just over eight months. 30
Who Might Benefit?
Optune is intended for treatment of glioblastoma in the supratentorial (or upper) region of the brain, not for tumours in the cerebellum or brain stem.26
Glioblastoma is more common in older people, and is more common in men than in women.31 Whereas primary glioblastoma most often affects older patients, secondary glioblastoma, which develops more slowly from a lower-grade glioma, usually occurs in younger patients.31
Precise numbers of Canadians with glioblastoma are not available, but based on estimates of the incidence of the disease from the US and Europe, approximately 1,200 Canadians may be diagnosed with glioblastoma each year.5,9,31 The Brain Tumour Foundation of Canada’s Canadian Brain Tumour Registry is collecting information on the incidence and survival rates for all types of brain cancers, and more comprehensive data should be available within the next few years.32
Current Practice
Patient care should be provided by a multidisciplinary team that includes specialists in neurosurgery, radiation oncology, neuropathology, neuro-oncology, and allied health.31
Standard treatment and management for glioblastoma include:
- surgical removal of as much of the tumour as possible while avoiding damage to critical areas of the brain, or biopsy only for inoperable tumours
- radiation therapy beginning three to six weeks after surgery and concurrent chemotherapy with oral temozolomide for three to six weeks
- subsequent maintenance temozolomide therapy for six cycles (or up to twelve months).4,9,11,31,33-35
Treatment may be modified for elderly or frail patients.34
Prognosis is affected by the patient’s age, Karnofsky Performance Status (which measures functional ability or disability), the amount of tumour that can be surgically removed, and the molecular features of the glioblastoma, such as genetic alterations associated with a better or worse response to temozolomide chemotherapy.4,31
Other drug therapies may be used to treat disease recurrence, but evidence that they improve survival is still not conclusive, and they may cause serious adverse events.31,36,37
The 2016 US National Comprehensive Cancer Network consensus-based guidelines include “lower-level” evidence recommendations for alternating electric fields therapy as an add-on treatment option to standard therapy for newly diagnosed patients with glioblastoma in the supratentorial region of the brain; and as a treatment option for patients with recurrent glioblastoma.3
Methods
These bulletins are not systematic reviews and do not involve a detailed critical appraisal. They are not intended to provide recommendations for or against a particular technology.
Study Selection
One author screened the literature search results and reviewed the full text of all potentially relevant studies. Studies were considered for inclusion if the intervention included alternating electric fields for glioblastoma. Conference abstracts and grey literature were included when they provided additional information to that available in the published studies.
Peer Review
A draft bulletin was reviewed by two clinical experts and the manufacturer.
Literature Search Strategy
A peer-reviewed literature search was conducted using the following bibliographic databases: MEDLINE, Embase, PubMed, and the Cochrane Library (2017, Issue 6). Grey literature was identified by searching relevant sections of the Grey Matters checklist (https://www.cadth.ca/grey-matters). No methodological filters were applied. The search was limited to English-language documents published between January 1, 2012 and May 30, 2017. Regular alerts updated the search until project completion; only citations retrieved before December 19, 2017 were incorporated into the analysis. Conference abstracts were retrieved through a search of the Embase database limited to the last two years.
The Evidence
Optune has been assessed in several clinical trials over the past decade;13 the most recent evidence includes two randomized controlled trials (EF-1430,38 and EF-1139) and the PRiDe Patient Registry Dataset.40
In the remainder of this section, the trials and the registry study are described first, followed by summaries of the main outcomes: overall survival, progression-free survival, compliance, quality-of-life, and safety. Smaller case series and subgroup analyses are also noted under the relevant outcomes sections.
Some trials included patients newly diagnosed with glioblastoma,30,38 whereas others included patients at first, second, or later recurrence of cancer.39 Consequently, the timing and duration of TTFields treatment varied.6,17 The drug therapies used in the trials also differed; for example, the use of temozolomide alone, or the use of triple therapy that included temozolomide and/or other drugs.30,38-40 The trials did not use a sham treatment in the control group, as it was considered impractical and inappropriate to place an additional burden on patients and caregivers (for example, shaving the patient’s head and placement of the transducer arrays.16,30,38
Most publications on Optune ― including studies, reviews, and commentaries ― received funding from the manufacturer.6-9,11,13,14,18,19,21,22,30,38-67
Study Characteristics
EF-14
Interim results from the EF-14 trial — the largest, multinational trial of TTFields therapy — were published in 2015,38 and the final study results were published in December 2017.30 The trial involved 695 patients at 83 centres. Patients were all newly diagnosed with glioblastoma and had all received standard treatment: debulking surgery (to reduce the size of the tumour), where feasible; radiation therapy; and chemotherapy. Participants were randomized at a ratio of 2 to 1 to receive maintenance therapy with either TTFields plus temozolomide (466 patients), or temozolomide only (229 patients). Patients in either group who experienced cancer progression could also receive second-line chemotherapy. Patients in the TTFields group who experienced cancer progression could continue to receive TTFields treatment. The trial excluded patients with disease progression prior to randomization. Patients who were not able to complete primary treatment and those who could not tolerate temozolomide chemotherapy were also excluded.
The interim analysis of EF-14 trial data was planned to occur when there was at least 18 months of follow-up for the first 315 randomized patients: 210 in the TTFields group, and 105 in the temozolomide-only group.38 Based on the positive interim results, the trial’s data and safety monitoring committee recommended stopping the trial, and TTFields therapy was offered to all patients.
Participants in the EF-14 trial were followed for a median of 40 months and a minimum of 24 months.30
EF-11
In the EF-11 trial, published in 2012, 237 patients with recurrent glioblastoma were randomized to receive either TTFields therapy only (n = 120) or the physician’s choice of chemotherapy (n = 117).39 To be included, patients must have received prior radiation therapy, with or without temozolomide chemotherapy. Patients with implanted medical devices, such as pacemakers, were excluded. Most patients were at the second or third recurrence of the disease. Their assigned treatment began within one week of randomization and continued until their disease progressed further or they were found to be unable to tolerate their assigned treatment. The trial was conducted at 28 centres in the US, Europe, and Israel.39
PRiDe
The Patient Registry Dataset (PRiDe) is a post-marketing registry study following 457 patients with recurrent glioblastoma who received the TTFields therapy in 91 US centres between October 2011 and November 2013.40 The intent was to gather real-world clinical practice information on patients treated with TTFields. The patients in the registry could have received any prior therapies and could have had any number of cancer recurrences. The median duration of TTField treatment in the registry patients was 4.1 months.40
Overall Survival
In the final analysis of the EF-14 trial, the median overall survival in the intention-to-treat patient groups was 20.9 months in the TTFields-plus-temozolomide group, and 16.0 months in the temozolomide-only group.30 More than half of the patients in both treatment groups also received second-line drug therapies: 67% of patients in the TTFields-plus-temozolomide group and 57% of those in the temozolomide-only group.38 At two years after trial enrolment, more of the patients in the TTFields-plus-temozolomide group were alive (43%) compared with the temozolomide-only group (31%).30
In the EF-11 trial of patients with disease recurrence, there was no difference in overall survival between the TTFields-only group and the chemotherapy-only group at any time point.39 Investigators concluded that the EF-11 trial showed that TTFields treatment offered comparable efficacy to that achieved with chemotherapy.59
Authors of the PRiDe registry study also conducted an analysis of survival. Median overall survival was 9.6 months.40 At one year, 44% of registry patients were alive, and at two years, 30% of patients were alive.40
A post-hoc analysis of the EF-11 trial found that median overall survival was higher in patients with recurrent glioblastoma who used the TTFields device for 18 or more hours daily (survival rate of 7.7 months) compared with patients who used the system less (survival rate of 4.5 months).47
Researchers at one US centre reported on a small case series that found improved survival in patients who received TTFields treatment in addition to triple-drug therapy (temozolomide, bevacizumab, and irinotecan).68 Median overall survival was 34.5 months for the 29 patients who received the combined treatment, and was slightly longer ― 39.4 months ― for the 15 patients who started TTFields treatment earlier (i.e., within 125 days of initial diagnosis).68
Another small study, reported in a conference abstract, found that the median overall survival was 12 months in 12 patients who received stereotactic radiosurgery in addition to TTFields therapy, compared with four months in 28 patients who received TTFields therapy without radiosurgery.67
A small case series of eight patients with bevacizumab-refractory glioblastoma reported a median overall survival of 7.2 months from the start of TTFields therapy.41 Several case reports mention long-term survivors of glioblastoma who have either continued to use TTFields therapy for several years (in addition to drug and other treatments),12,47,55,69 or who have been in good health and are no longer receiving treatment after receiving TTFields treatment.47,49,56,70
Progression-Free Survival
Progression-free survival is the period of time during and after treatment in which the disease does not worsen. Final EF-14 trial results reported that median progression-free survival in the intention-to-treat patient groups was 6.7 months in the TTFields-plus-temozolomide group, and 4.0 months in the temozolomide-only group.30
There was no difference in median progression-free survival at six months in the EF-11 trial patients with recurrent glioblastoma (just over two months in both the TTFields-only group and the chemotherapy-only group).39
Compliance With Treatment
The use of TTFields for 18 or more hours per day is associated with better overall survival.6,7,30,47,71 At least one continuous 28-day cycle of TTFields treatment is necessary to be able to see a treatment effect (usually based on MRI imaging, if available).47,48 The Optune system records the amount of time the patient uses the device in a monthly report for the attending physician.18
In the EF-14 trial, 75% of the patients in the TTFields-plus-temozolomide group wore the device for an average of 18 or more hours per day.30
In the EF-11 trial, of the 116 patients who started TTFields treatment, 78% completed at least one four-week cycle.39 The average compliance with treatment was 86% (an average of 20.6 hours per day).39 Twenty-seven patients stopped TTFields treatment early, either because of not using the system for the recommended hours each day or because of an inability to manage the device.39
PRiDe registry compliance data were available for only about two-thirds of the patients (n = 287).40 About 44% of the patients (n = 127) used the TTFields therapy for 18 or more hours per day. Patients with more functional impairment were less compliant in their use of the TTFields treatment.40
A small case series of patients with bevacizumab-refractory glioblastoma reported a compliance rate of 74.2%; this was based on seven of the eight patients’ records, with a median duration of TTFields therapy of 5.2 months.41
Quality of Life
Interim EF-14 trial results showed an initial improvement in quality of life in the patients treated with TTFields plus temozolomide, but at the nine-month follow-up there was no difference between the patient groups.43 Self-reported physical and social functioning were also similar between the groups at the nine-month and the 12-month follow-up.43 Final EF-14 study results on quality of life are forthcoming.30
Data on quality of life was available for 63 patients (27%) in the EF-11 trial who remained on TTFields therapy for at least three months.39 These patients reported a higher quality of life — including better cognitive and emotional status — than those in the chemotherapy group, possibly because they were not experiencing the toxic effects of chemotherapy.11 There was no difference between the groups in the domains of overall health and social functioning.39
Safety
Skin Irritation
The most common adverse event with the use of TTFields therapy is skin irritation, including rash, ulceration, and infections.19,21,72 The irritation is mainly due to repeated shaving of the scalp, application and removal of the transducer arrays, allergic reaction to the hydrogel, and the pressure of the transducers on the skin.14,19 Skin irritation was usually mild to moderate, and it was treatable with topical steroids or antibiotics and by slightly adjusting the positioning of the transducer arrays and adhesives when they were exchanged. It was also treated by removing the arrays for a few days.19,72
In the EF-14 trial, about 52% of patients in the TTFields group had mild to moderate skin reactions, and about 2% of patients had more severe skin reactions.30 Patients in the TTFields-plus-temozolomide treatment group reported “itchy skin” more often than those in the temozolomide-only group.43 In the EF-11 trial, 16% of patients in the TTFields group experienced mild to moderate skin irritation.39
In the PRiDe registry, 24.3% of patients (the actual patient numbers were not reported) experienced skin reactions.40 Although some patients reported a sensation of scalp warmth, this did not cause injury.40 (When the device is in operation, sensors in the arrays monitor temperature. The system sounds an alarm and automatically shuts off if the temperature rises above 105.8oF or 41oC.19)
Systemic Adverse Events
In the EF-14 trial, systemic adverse events such as blood and gastrointestinal disorders, seizures, and infections were similar in the patients who received TTFields plus temozolomide, and those who received temozolomide only.30 In the EF-11 trial, systemic adverse events such as gastrointestinal and blood disorders, and infections, occurred more frequently in the chemotherapy patient group.39
A post hoc analysis of 144 patients in the EF-14 trial who received TTFields in addition to second-line drug therapies after disease recurrence found a higher incidence of adverse events compared with patients who received chemotherapy only.51 These results may have been confounded by the second-line drug therapies used (most commonly bevacizumab), the longer follow-up period for patients who received TTFields, and the 14 patients who crossed over to the TTFields treatment arm who had not initially received TTFields.51
Other Adverse Events
Other adverse events reported in the registry patients included neurological disorder (10.4%), seizure (8.9%), headache (5.7%), pain or discomfort (4.7%), and falls (3.9%). Actual patient numbers were not reported.40
In the EF-14 study, the rates of seizures were similar in the patients who received TTFields plus temozolomide and those who received temozolomide only.30
TTFields therapy should not be used in patients with active implanted devices — such as deep brain, spinal cord, or vagal nerve stimulators; or pacemakers — or in individuals with a sensitivity to the conductive hydrogels.25,26,61 A 2017 laboratory study concluded that radiation therapy should not be used when the transducer arrays are in place because of an increased risk of skin toxicity.50
Cost Studies
No Canadian economic evaluations were identified.
French researchers examined the cost-effectiveness of TTFields therapy in patients newly diagnosed with glioblastoma based on interim data from the EF-14 trial.29 Two scenarios were considered: the first, standard of care alone, which included radiation therapy and temozolomide; and the second, TTFields treatment plus standard of care. With a projected life expectancy of 22.08 months in the TTFields scenario and 18 months in the standard of care alone scenario, TTFields therapy costs €243,141 compared with €57,665 for standard treatment alone; with an incremental cost-effectiveness ratio (ICER) of €549,909 per life-year gained.29 A Swedish cost-effectiveness evaluation using more recent patient data has also been conducted; however, the results are not available in English.73
A systematic review of economic studies assessing the cost-effectiveness of glioblastoma therapies, including TTFields therapy, is underway at the University of York and expected to be published in 2018.74
Concurrent Developments
Treatments for Glioblastoma
Molecular-based tumour typing is increasingly important in cancer diagnosis and treatment decisions.75,76 People with glioblastoma tumours that have particular genetic alterations have better prognoses than those with tumours having only some or none of these alterations.75
Many clinical trials for glioblastoma therapies are in various phases of development.77-79 Some researchers have speculated there may be a synergistic effect between TTFields therapy and the response to drug therapies.42,57,62 Clinical trials of TTFields therapy in combination with other therapies are also in progress.8,17,80
Immunotherapies, vaccines, and oncologic viruses that promote the patient’s immune system response or target tumour cells are other areas of investigation.76,77,81-83 Glioblastoma is one of the cancers included in chimeric antigen receptor (CAR) T-cell therapy research — where a patient’s T-cells are extracted, genetically modified, and reinfused to attack cancer cells.78,84,85
Ultrasound technologies, such as microbubbles that temporarily allow better access of drug therapies across the blood-brain barrier, and focused ultrasound ablation (or removal) of tumours have also been investigated in the treatment of glioblastoma.5,86,87
Another device-based treatment for glioblastoma is the Nativis Voyager (Nativis, Seattle), which is undergoing feasibility and safety trials in the US and Australia.88,89 The Nativis Voyager is worn on a headband and the device delivers ultra-low radiofrequency energy to the brain. The radiofrequency energy is believed to disrupt genetic processes in the cancer cells — in particular, the epidermal growth factor receptor — and prevent cell division.89
Other Uses of Tumour-Treating Fields
Small studies of TTFields in treating other types of cancers have also been published or are underway, including investigations of its use in meningioma — a brain tumour that forms on the membranes covering the brain and spinal cord; brain metastases from other types of cancer; mesothelioma (a rare cancer related to asbestos exposure); and pancreatic, lung, and ovarian cancers.8,11,13,17,36 A Danish trial is investigating the feasibility of increasing TTField intensity to the tumour by craniectomy ― removing small pieces of the overlaying skull.52
A report of compassionate use of TTFields treatment in five children and adolescents with high grade gliomas (including glioblastoma) concluded a pediatric trial would be worthwhile,90 and the Pediatric Brain Tumor Consortium is conducting a trial in children.91
Implementation Issues
Uptake
A 2017 US review noted that uptake of TTFields treatment has been relatively slow, and that only about 15% of patients newly diagnosed with glioblastoma were treated with TTFields in 2016.7 Worldwide, over 5,000 patients with glioblastoma have been treated with TTFields (Justin Kelly, Novocure, Portsmouth, NH: personal communication, 2017 Oct 30).
Training Needs
Originally, Novocure clinical staff customized the layout of the transducer arrays for each patient.92 Now, physicians who prescribe Optune must receive training and certification to do this for their patients using the NovoTAL System.33,92,93 A study involving 14 physicians (neuro-oncologists, medical oncologists, and neurosurgeons) certified to prescribe Optune found that physicians in all specialties were able to use the system to accurately initiate and adjust patient treatment.92
Patients and their families or caregivers also need training in using the device and in scalp preparation, and in ensuring the correct placement of the transducer arrays.1,7,14
Caregiver Burden
Cleansing and shaving the scalp, replacing the electrodes every three days, and wearing the device for most of the day and night may be a burden for some patients.7 Most patients will need help to prepare their scalp and apply the arrays, and family or caregiver support will likely be needed.7,22,59
Patient Perspective
The need to shave the head and wear the noticeable transducer arrays and wires may be seen as a stigma of cancer, as with hair loss in chemotherapy.7,16,63,65,94
Jeffrey Weiss, a US journalist with glioblastoma who has since passed away, chronicled his experience as a patient with this disease, including his experience using the TTFields therapy:
“Optune use is a real-world physical hassle. Four palm-sized patches of electrodes are stuck to my scalp in a particular pattern. Every two days, the electrodes must be removed, my scalp cleaned and reshaved, and a new set of electrodes stuck on. (My wife, Marni, has to do the electrode installation; I can’t possibly reach all around my head to get them on correctly.) Sweat is a problem. It can make the electrodes fall off. Heat is a problem; if my head gets even a bit warm, an alarm goes off. When I leave the house, I wear hats to cover the odd-looking electrode patches…”95,96
One investigator with the EF-14 study noted that patients:
“…became psychologically dependent on the TTFields device, and saw it as a tangible way to treat their own disease. They were responsible for wearing the device, taking it with them throughout the day, and recharging the batteries. They also had to change their transducer arrays every few days…”61
This is in contrast to other cancer treatments where patients are more passive recipients of care.22,61 As one US patient who has used the system for two years said: “I wear it and wear it proudly… It’s an incredible machine and I’m fine not having hair.”97
Some patients found the system cumbersome, but future design modifications are expected.8,62 A 2016 conference abstract, reporting one centre’s experience with providing TTFields therapy, described the following reasons patients gave for not using the system: inability to tolerate the side effects (not specified), lack of caregiver support, and impact on lifestyle.98 In addition, two patients did not start TTFields therapy as prescribed because of social or financial reasons.98
Patient Education
Health care providers will need to educate patients and their families about the importance of compliance, as the effectiveness of TTFields depends on the device being worn and turned on.7,22,47 A recent guide for nursing staff noted that some patients experience cognitive impairment as a result of their disease treatments, so that user errors ― such as not noticing the device was not turned back on after a break in therapy ― may occur, which can affect compliance.22 The device’s compliance report for physicians may help to identify these types of issues.22 In addition to supporting the health care professionals, Novocure technicians are available to provide technical support to patients and caregivers.22
Patient Selection
It is not yet clear which patients with glioblastoma are most likely to benefit from TTFields treatment.16,17 The distribution of the electrical fields to the tumour is affected by many variables in addition to the positioning of the transducer arrays, including the location and shape of the tumour and whether it contains a core of necrotic (or dead) tissue, the layer of cerebrospinal fluid, and the insulating properties of adjacent tissue.99
Preliminary evidence suggests that patients who may benefit most are those:
- with tumours that are more superficial, rather than those located deep within the brain.100
- who have not previously received bevacizumab. These patients may have a better prognosis.40,64 However, a small subgroup analysis also suggested that patients who have developed resistance to bevacizumab may have a better response to TTFields therapy — possibly because of an altered immune response after receiving bevacizumab.47
- with tumours that developed from lower grade gliomas. These tumours appear to have a better response to TTFields therapy.45 As primary and secondary glioblastomas differ genetically, in the future, the genetic profiling of tumours may allow for better identification of patients who will benefit from TTFields treatment.45
Imaging Needs
Planning the patient’s transducer array layout is based on existing MRI imaging taken as part of standard care for glioblastoma (Justin Kelly, Novocure, Portsmouth, NH: personal communication, 2017 Oct 30). Subsequent MRIs, every two to three months, are also part of standard follow-up for patients with glioblastoma.14 For a more accurate assessment of treatment response in glioblastoma, investigators are exploring advanced MRI-based techniques, such as diffusion tensor imaging, 3-D echo-planar spectroscopy, and dynamic susceptibility contrast perfusion-weighted imaging.54,101
Pseudoprogression
In patients with glioblastoma, pseudoprogression (transient visual changes in the tumour not indicative of true disease progression), commonly due to tissue necrosis from radiation therapy, is often seen on follow-up MRIs within the first months after chemotherapy and radiation treatment.14,31,102 There is also a delay between treatment response with TTFields and when this becomes evident on MRI images.14 Continuation of TTFields treatment is recommended, regardless of early diagnostic imaging results.7,48,49,70,93 The effects of a prolonged use of TTFields after cancer recurrence — for example, on slowing the rate of progression or on tumour regression — warrant further investigation.16
Other Issues
The cost of TTFields therapy will be a barrier for patients if this therapy is not covered by public and private health insurance.7,23
The reduced incidence of systemic adverse events with TTFields therapy for patients with recurrent glioblastoma may reduce health care costs associated with treating more serious adverse events caused by chemotherapy and radiation therapy.8 A 2012 Australian health technology assessment of TTFields therapy included unpublished manufacturer’s information that found fewer hospitalizations and reduced length of stay among the patients who received TTFields treatment compared with those who received best standard chemotherapy.103
In the US, the Novocure support technicians meet with patients each month to provide support and download the compliance report for the health care team.22 How this works for patients outside of the US is not clear.
Final Remarks
In September 2017, Novocure announced that the German Federal Joint Committee, the Gemeinsamer Bundesausschuss, will jointly fund a new trial to assess the benefit of starting TTFields treatment earlier — as part of initial therapy alongside radiation therapy and temozolomide.104
Treatments that may improve survival without negatively affecting quality of life are important for patients with this aggressive, difficult-to-treat cancer.43 Nevertheless, some clinicians remain skeptical about the benefits of TTFields — perhaps because it is so different from traditional cancer treatments.7,16,23,63 Further research is underway that may clarify the place of TTFields in the treatment of glioblastoma.
References
- Optune® patient information and operation manual [Internet]. Jersey (GB): Novocure; 2017 May. [cited 2017 Sep 20]. Available from: https://www.optune.com/Content/pdfs/Optune_PIOM_8.5x11.pdf
- Brain tumour facts [Internet]. London (ON): Brain Tumour Foundation of Canada; 2017. [cited 2017 Aug 31]. Available from: http://www.braintumour.ca/2494/brain-tumour-facts
- National Comprehensive Cancer Network. Central nervous system cancers [Internet]. Version 1.2017. Fort Washington (PA): NCCN; 2017 Aug 18. [cited 2017 Dec 4]. (NCCN clinical practice guidelines in oncology). Available from: https://www.nccn.org/professionals/physician_gls/pdf/cns.pdf Registration required.
- Toms SA, Tapinos N. Recent advances in the treatment of gliomas - comprehensive brain tumor center. R I Med J. 2017 Jun 1;100(6):43-6.
- Gerstel J. Rethinking the brain and glioblastoma. Globe and Mail [Internet]. 2017 Apr 26 [cited 2017 Aug 28]. Available from: https://www.theglobeandmail.com/life/health-and-fitness/advsunnybrook/rethinking-the-brain-and-glioblastoma/article34664258/
- Mrugala MM, Ruzevick J, Zlomanczuk P, Lukas RV. Tumor treating fields in neuro-oncological practice. Curr Oncol Rep. 2017 Aug;19(8):53.
- Burri SH, Gondi V, Brown PD, Mehta MP. The evolving role of tumor treating fields in managing glioblastoma: guide for oncologists. Am J Clin Oncol. 2017 Aug 24. Epub ahead of print.
- Mittal S, Klinger NV, Michelhaugh SK, Barger GR, Pannullo SC, Juhasz C. Alternating electric tumor treating fields for treatment of glioblastoma: rationale, preclinical, and clinical studies. J Neurosurg. 2017 Feb 24;1-8. Epub ahead of print.
- Davis ME. Glioblastoma: overview of disease and treatment. Clin J Oncol Nurs [Internet]. 2016 Oct 1 [cited 2017 Jul 20];20(5):S2-S8. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5123811/pdf/nihms-820394.pdf
- Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005 Mar 10;352(10):987-96.
- Mehta M, Wen P, Nishikawa R, Reardon D, Peters K. Critical review of the addition of tumor treating fields (TTFields) to the existing standard of care for newly diagnosed glioblastoma patients. Crit Rev Oncol Hematol [Internet]. 2017 Mar [cited 2017 Jul 20];111:60-5. Available from: http://www.sciencedirect.com/science/article/pii/S1040842817300070?via%3Dihub
- Grady D. Electrical scalp device can slow progression of deadly brain tumors. N Y Times [Internet]. 2014 Nov 15 [cited 2017 Aug 31]. Available from: https://www.nytimes.com/2014/11/16/health/electrical-scalp-device-can-slow-progression-of-deadly-brain-tumors.html
- Bomzon Z, Wenger C. Of fields and phantoms: the importance of virtual humans in optimizing cancer treatment with tumor treating fields. IEEE Pulse. 2017 Jul;8(4):46-9.
- Trusheim J, Dunbar E, Battiste J, Iwamoto F, Mohile N, Damek D, et al. A state-of-the-art review and guidelines for tumor treating fields treatment planning and patient follow-up in glioblastoma. CNS Oncol [Internet]. 2017 Jan [cited 2017 Jul 20];6(1):29-43. Available from: https://www.futuremedicine.com/doi/pdf/10.2217/cns-2016-0032
- Sampson JH. Alternating electric fields for the treatment of glioblastoma. JAMA. 2015 Dec 15;314(23):2511-3.
- Cloughesy TF, Lassman AB. NovoTTF: where to go from here? Neuro Oncol. 2017 May 1;19(5):605-8.
- Hottinger AF, Pacheco P, Stupp R. Tumor treating fields: a novel treatment modality and its use in brain tumors. Neuro Oncol [Internet]. 2016 Oct [cited 2017 Jul 20];18(10):1338-49. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5035531/pdf/now182.pdf
- Wong ET. Clinical use of tumor treating fields and other new technologies in cancer. Clin Adv Hematol Oncol. 2015 Nov;13(11 Suppl 11):8-12.
- Lacouture ME, Davis ME, Elzinga G, Butowski N, Tran D, Villano JL, et al. Characterization and management of dermatologic adverse events with the NovoTTF-100A System, a novel anti-mitotic electric field device for the treatment of recurrent glioblastoma. Semin Oncol [Internet]. 2014 Jun [cited 2017 Jul 20];41(suppl 4):s1-14. Available from: http://www.sciencedirect.com/science/article/pii/S0093775414000591
- Kinzel A, Ambrogi M, Varshaver M, Benson L, Kirson E. First report of user experience, compliance outcomes, and alarm states with the second generation Optune system. Neuro Oncol [Internet]. 2016 [cited 2017 Jul 20];18(suppl 6):vi135. Available from: https://academic.oup.com/neuro-oncology/article-abstract/18/suppl_6/vi135/2542899/ (Presented at The 21st Annual Scientific Meeting and Education Day of the Society for Neuro-Oncology; 2016 Nov 17-20; Scottsdale, AZ).
- Swanson KD, Lok E, Wong ET. An overview of alternating electric fields therapy (NovoTTF Therapy) for the treatment of malignant glioma. Curr Neurol Neurosci Rep [Internet]. 2016 Jan [cited 2017 Jul 20];16(1):8. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4703612/pdf/11910_2015_Article_606.pdf
- Murphy J, Bowers ME, Barron L. Optune: practical nursing applications. Clin J Oncol Nurs [Internet]. 2016 Oct 1 [cited 2017 Jul 20];20(5):S14-S19. Available from: https://cjon.ons.org/cjon/20/5/supplement/optune-practical-nursing-applications
- Chamberlain MC. Treatment for patients with newly diagnosed glioblastoma. JAMA. 2016 Jun 7;315(21):2348.
- NovoCure Ltd. Effect of NovoTTF-100A together with temozolomide in newly diagnosed glioblastoma multiforme (GBM). 2009 Jun 9 [cited 2017 Oct 18; updated 2017 Apr 10]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine; 2000 - . Available from: https://www.clinicaltrials.gov/ct2/show/NCT00916409 NLM Identifier: NTC00916409.
- Press release, FDA approves expanded indication for medical device to treat a form of brain cancer [Internet]. Silver Spring (MD): U.S. Food and Drug Administration; 2015 Oct 5. [cited 2017 Sep 20]. Available from: https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm465744.htm
- Summary of safety and effectiveness data (SSED) [Internet]. Silver Spring (MD): U.S. Food and Drug Administration; 2015 Oct 5. [cited 2017 Sep 20]. Available from: https://www.accessdata.fda.gov/cdrh_docs/pdf10/P100034S013B.pdf
- Novocure announces commercial launch of NovoTTF™ therapy in Europe and Israel [Internet]. St. Helier (Jersey Isle): Novocure Limited; 2014 Jul 15. [cited 2017 Oct 31]. Available from: https://s21.q4cdn.com/825405078/files/doc_news/2014/eu-commercial-launch-final-071414.pdf
- Tumor treating fields therapy (Optune) for recurrent glioblastoma. Plymouth Meeting (PA): ECRI Institute; 2016. (Emerging technology evidence report).
- Bernard-Arnoux F, Lamure M, Ducray F, Aulagner G, Honnorat J, Armoiry X. The cost-effectiveness of tumor-treating fields therapy in patients with newly diagnosed glioblastoma. Neuro Oncol. 2016 Aug;18(8):1129-36.
- Stupp R, Taillibert S, Kanner A, Read W, Steinberg DM, Lhermitte B, et al. Effect of tumor-treating fields plus maintenance temozolomide vs maintenance temozolomide alone on survival in patients with glioblastoma: a randomized clinical trial. JAMA. 2017;318(23):2306-16.
- Easaw JC, Mason WP, Perry J, Laperriere N, Eisenstat DD, Del MR, et al. Canadian recommendations for the treatment of recurrent or progressive glioblastoma multiforme. Curr Oncol [Internet]. 2011 Jun [cited 2017 Sep 22];18(3):e126-e136. Available from: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3108873
- Canadian brain tumour registry [Internet]. London (ON): Brain Tumour Foundation of Canada; 2017. [cited 2017 Aug 31]. Available from: http://www.braintumour.ca/4475/brain-tumour-registry
- Batchelor T, Loeffler JS, Wen PY, Eichler AF. Initial postoperative therapy for glioblastoma and anaplastic astrocytoma. In: Post TW, editor. UpToDate [Internet]. Waltham (MA): UpToDate; 2016 Dec 8 [cited 2017 Aug 29]. Available from: www.uptodate.com Subscription required.
- Sulman EP, Ismaila N, Armstrong TS, Tsien C, Batchelor TT, Cloughesy T, et al. Radiation therapy for glioblastoma: American Society of Clinical Oncology clinical practice guideline endorsement of the American Society for Radiation Oncology guideline. J Clin Oncol [Internet]. 2017 [cited 2017 Jul 24];35(3):361-9. Available from: http://ascopubs.org/doi/pdfdirect/10.1200/JCO.2016.70.7562
- Perry JR, Laperriere N, O’Callaghan CJ, Brandes AA, Menten J, Phillips C, et al. Short-course radiation plus temozolomide in elderly patients with glioblastoma. N Engl J Med. 2017 Mar 16;376(11):1027-37.
- Hays, Inc. Novocure (tumor treating fields). Updated final evidence report [Internet]. Olympia (WA): Washington State Health Care Authority, Health Technology Assessment Program; 2015 Dec 26. [cited 2017 Sep 22]. Available from: https://www.hca.wa.gov/assets/program/novocure_final_report_121015%5b1%5d.pdf
- Seystahl K, Gramatzki D, Roth P, Weller M. Pharmacotherapies for the treatment of glioblastoma - current evidence and perspectives. Expert Opin Pharmacother. 2016 Jun;17(9):1259-70.
- Stupp R, Taillibert S, Kanner AA, Kesari S, Steinberg DM, Toms SA, et al. Maintenance therapy with tumor-treating fields plus temozolomide vs temozolomide alone for glioblastoma: a randomized clinical trial. JAMA. 2015 Dec 15;314(23):2535-43.
- Stupp R, Wong ET, Kanner AA, Steinberg D, Engelhard H, Heidecke V, et al. NovoTTF-100A versus physician’s choice chemotherapy in recurrent glioblastoma: a randomised phase III trial of a novel treatment modality. Eur J Cancer [Internet]. 2012 Sep [cited 2017 Jul 20];48(14):2192-202. Available from: http://www.sciencedirect.com/science/article/pii/S0959804912003528?via%3Dihub
- Mrugala MM, Engelhard HH, Tran DD, Kew Y, Cavaliere R, Villano JL, et al. Clinical practice experience with NovoTTF-100ATM system for glioblastoma: the Patient Registry Dataset (PRiDe). Semin Oncol [Internet]. 2015 [cited 2017 Jul 20];42(3):e33. Available from: http://www.sciencedirect.com/science/article/pii/S0093775415000561 Erratum for: Seminars in Oncology. 2014;41:(5):S4-S13.
- Ansstas G, Tran DD. Treatment with tumor-treating fields therapy and pulse dose bevacizumab in patients with bevacizumab-refractory recurrent glioblastoma: a case series. Case Rep Neurol [Internet]. 2016 Jan [cited 2017 Jul 20];8(1):1-9. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4748800/pdf/crn-0008-0001.pdf
- Wong ET, Lok E, Swanson KD. Clinical benefit in recurrent glioblastoma from adjuvant NovoTTF-100A and TCCC after temozolomide and bevacizumab failure: a preliminary observation. Cancer Med [Internet]. 2015 Mar [cited 2017 Jul 20];4(3):383-91. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4380964/pdf/cam40004-0383.pdf
- Zhu JJ, Demireva P, Kanner AA, Pannullo S, Mehdorn M, Avgeropoulos N, et al. Health-related quality of life, cognitive screening, and functional status in a randomized phase III trial (EF-14) of tumor treating fields with temozolomide compared to temozolomide alone in newly diagnosed glioblastoma. J Neurooncol [Internet]. 2017 Aug 28 [cited 2017 Sep 7]. Available from: https://link.springer.com/content/pdf/10.1007%2Fs11060-017-2601-y.pdf
- Turner SG, Gergel T, Wu H, Lacroix M, Toms SA. The effect of field strength on glioblastoma multiforme response in patients treated with the NovoTTFTM-100A system. World J Surg Oncol [Internet]. 2014 May 22 [cited 2017 Jul 20];12:162. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4036833/pdf/1477-7819-12-162.pdf
- Wong ET, Lok E, Swanson KD, Gautam S, Engelhard HH, Lieberman F, et al. Response assessment of NovoTTF-100A versus best physician’s choice chemotherapy in recurrent glioblastoma. Cancer Med [Internet]. 2014 Jun [cited 2017 Jul 20];3(3):592-602. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4101750/pdf/cam40003-0592.pdf
- Omar AI. Tumor treating field therapy in combination with bevacizumab for the treatment of recurrent glioblastoma. J Vis Exp [Internet]. 2014 Oct 27 [cited 2017 Jul 20];(92):e51638. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4541554/pdf/jove-92-51638.pdf
- Kanner AA, Wong ET, Villano JL, Ram Z. Post hoc analyses of intention-to-treat population in Phase III comparison of NovoTTF-100ATM system versus best physician’s choice chemotherapy. Semin Oncol [Internet]. 2015 [cited 2017 Jul 20];42(3):e56. Available from: http://www.sciencedirect.com/science/article/pii/S0093775415000548 Erratum for: Seminars in Oncology (2014) 41(5):S25-S34.
- Vymazal J, Wong ET. Response patterns of recurrent glioblastomas treated with tumor-treating fields. Semin Oncol. 2015;42(3):e44. Erratum for: Seminars in Oncology. 2014;41(5):S14-S24.
- Rulseh AM, Keller J, Klener J, Sroubek J, Dbaly V, Syrucek M, et al. Long-term survival of patients suffering from glioblastoma multiforme treated with tumor-treating fields. World J Surg Oncol [Internet]. 2012 Oct 24 [cited 2017 Jul 20];10:220. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3514151/pdf/1477-7819-10-220.pdf
- Bender E, Kozak K, Howard S, Hayes L, Bayouth J, Robins HI. The effect of OptuneTM tumor treating fields transducer arrays on skin radiation dose during radiotherapy. J Clin Neurosci. 2017 Apr 17;42:172-5.
- Kesari S, Ram Z, Trial I. Tumor-treating fields plus chemotherapy versus chemotherapy alone for glioblastoma at first recurrence: a post hoc analysis of the EF-14 trial. CNS Oncol [Internet]. 2017 Apr 12 [cited 2017 Jul 20]. Available from: https://www.futuremedicine.com/doi/pdf/10.2217/cns-2016-0049
- Korshoej AR, Saturnino GB, Rasmussen LK, Von OG, Sorensen JC, Thielscher A. Enhancing predicted efficacy of tumor treating fields therapy of glioblastoma using targeted surgical craniectomy: a computer modeling study. PLoS One [Internet]. 2016 [cited 2017 Jul 20];11(10):e0164051. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5047456/pdf/pone.0164051.pdf
- Schwartz MA, Onuselogu L. Rationale and background on tumor-treating fields for glioblastoma. Clin J Oncol Nurs [Internet]. 2016 Oct 1 [cited 2017 Jul 20];20(5):S20-S24. Available from: https://cjon.ons.org/cjon/20/5/supplement/rationale-and-background-tumor-treating-fields-glioblastoma
- Connelly J, Hormigo A, Mohilie N, Hu J, Chaudhry A, Blondin N. Planning TTFields treatment using the NovoTAL system-clinical case series beyond the use of MRI contrast enhancement. BMC Cancer [Internet]. 2016 Nov 4 [cited 2017 Jul 20];16(1):842. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5096303/pdf/12885_2016_Article_2890.pdf
- Villano JL, Williams LE, Watson KS, Ignatius N, Wilson MT, Valyi-Nagy T, et al. Delayed response and survival from NovoTTF-100A in recurrent GBM. Med Oncol. 2013 Mar;30(1):338.
- Kirson ED, Dbaly VF, Tovarys FF, Vymazal JF, Soustiel JF FAU - Itzhaki A, Itzhaki AF, et al. Alternating electric fields arrest cell proliferation in animal tumor models and human brain tumors. Proc Natl Acad Sci U S A [Internet]. 2007 [cited 2017 Sep 22];104(24):10152-7. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1886002/
- Kirson ED, Schneiderman RS, Dbaly V, Tovarys F, Vymazal J, Itzhaki A, et al. Chemotherapeutic treatment efficacy and sensitivity are increased by adjuvant alternating electric fields (TTFields). BMC Med Phys [Internet]. 2009 Jan 8 [cited 2017 Sep 22];9:1. Available from: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2647898
- Davies AM, Weinberg U, Palti Y. Tumor treating fields: a new frontier in cancer therapy. Ann N Y Acad Sci. 2013 Jul;1291:86-95.
- Fonkem E, Wong ET. NovoTTF-100A: a new treatment modality for recurrent glioblastoma. Expert Rev Neurother. 2012 Aug;12(8):895-9.
- Saria MG, Kesari S. Efficacy and safety of treating glioblastoma with tumor-treating fields therapy. Clin J Oncol Nurs [Internet]. 2016 Oct 1 [cited 2017 Jul 20];20(5):S9-S13. Available from: https://cjon.ons.org/cjon/20/5/supplement/efficacy-and-safety-treating-glioblastoma-tumor-treating-fields-therapy
- Toms SA. Tumor treating fields and other technological advances in the treatment of cancer. Clin Adv Hematol Oncol. 2015 Nov;13(11 Suppl 11):3-7.
- Zhu P, Zhu JJ. Tumor treating fields: a novel and effective therapy for glioblastoma: mechanism, efficacy, safety and future perspectives. Chin Clin Oncol [Internet]. 2017 Aug [cited 2017 Sep 7];6(4):41. Available from: http://cco.amegroups.com/article/view/16215/16369
- Wick W. TTFields: where does all the skepticism come from? Neuro Oncol [Internet]. 2016 Mar [cited 2017 Jul 20];18(3):303-5. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4767251/pdf/now012.pdf
- Wong ET, Lok E, Swanson KD. An evidence-based review of alternating electric fields therapy for malignant gliomas. Curr Treat Options Oncol [Internet]. 2015 Aug [cited 2017 Jul 20];16(8):40. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4491358/pdf/11864_2015_Article_353.pdf
- Butowski N, Wong ET, Mehta MP, Wilson LK. A roundtable discussion on the clinical challenges and options for the treatment of glioblastoma: introducing a novel modality, TTFields. Semin Oncol. 2013 Dec;40(6):S2-S4.
- Zhu JJ, Pannullo S, Mehdorn M, Payer F, Avgeropoulos N, Salmaggi A, et al. Quality of life, cognitive function and functional status in the EF-14 trial: a prospective, multi-center trial of TTfields with temozolomide compared to temozolomide alone in patients with newly diagnosed GBM. Neuro Oncol [Internet]. 2015 [cited 2017 Jul 20];17(suppl 5):v9. Available from: https://academic.oup.com/neuro-oncology/article/17/suppl_5/v9/2765417/ (Presented at The 20th Annual Scientific Meeting of the Society for Neuro-Oncology; 2015 Nov 19-22; San Antonio, TX).
- Mahadevan A, Floyd SR, Wong ET, Kasper E, Barron L. Survival benefit of NovoTTF therapy plus stereotactic radiosurgery for recurrent malignant gliomas. Int J Radiat Oncol Biol Phys [Internet]. 2015 [cited 2017 Jul 21];93(3 Suppl 1):E58. Available from: http://www.redjournal.org/article/S0360-3016(15)01420-0/pdf
- Lu G, Rao M, Delumpa LC, Liu Z, Hsu S, Zhu JJ. Survival benefit with triple chemotherapy and TTFields for glioblastoma. Neuro Oncol [Internet]. 2016 [cited 2017 Jul 20];18(Suppl 6):vi149. Available from: https://academic.oup.com/neuro-oncology/article-abstract/18/suppl_6/vi149/2543015/ (Presented at The 21st Annual Scientific Meeting and Education Day of the Society for Neuro-Oncology; 2016 Nov 17-20; Scottsdale, AZ).
- Meletath SK, Pavlick D, Brennan T, Hamilton R, Chmielecki J, Elvin JA, et al. Personalized treatment for a patient with a BRAF V600E mutation using dabrafenib and a tumor treatment fields device in a high-grade glioma arising from ganglioglioma. J Natl Compr Canc Netw. 2016 Nov;14(11):1345-50.
- Rulseh A, Sroubek J, Klener J, Vymazal J. Longterm survival in glioblastoma patients after tumor treating fields (TTFields) therapy. Cancer Res. 2017;77(13 Suppl 1):CT141. (Presented at American Association for Cancer Research Annual Meeting 2017; 2017 Apr 1-5; Washington, DC).
- Stupp R, Hegi ME, Idbaih A, Steinberg D, Lhermitte B, Read W, et al. Tumor treating fields added to standard chemotherapy in newly diagnosed glioblastoma (GBM): final results of a randomized, multi-center, phase III trial. Cancer Res [Internet]. 2017 Jul [cited 2017 Sep 18];77(13 (Supplement)):CT007. Available from: http://cancerres.aacrjournals.org/content/77/13_Supplement/CT007 (Presented at AACR Annual Meeting 2017; April 1-5, 2017; Washington, DC).
- Lukas RV, Ratermann KL, Wong ET, Villano JL. Skin toxicities associated with tumor treating fields: case based review. J Neurooncol. 2017 Aug 28. Epub ahead of print.
- Optune: Medicinteknisk produkt som genererar “tumor treating fields” för behandling av glioblastom [Internet]. Stockholm (SE): Tandvårds- och läkemedelsförmånsverket (TLV); 2017 Oct 3. [cited 2017 Nov 2]. Available from: https://www.tlv.se/Upload/Medicinteknik/halsoekonomisk_utvardering_optune.pdf Swedish.
- A systematic review of economic analyses used to assess the cost-effectiveness of health technologies in the management of glioblastoma. PROSPERO: International prospective register of systematic reviews [Internet]. 2017 [cited 2017 Aug 31];CRD42017053394. Available from: https://www.crd.york.ac.uk/PROSPERO/display_record.asp?ID=CRD42017053394
- Uhm JH, Porter AB. Treatment of glioma in the 21st century: an exciting decade of postsurgical treatment advances in the molecular era. Mayo Clin Proc. 2017 Jun;92(6):995-1004.
- Morgan E, Mason W. What are the prospects for combination therapy for glioblastoma? Expert Rev Neurother. 2017 Jul 12;1-3.
- Breindl A. Making lemonade: Zika virus kills glioblastoma stem cells. BioWorld MedTech. 2017 Sep 21;25(183):1, 7.
- Domingo-Musibay E, Galanis E. What next for newly diagnosed glioblastoma? Fut Oncol [Internet]. 2015 [cited 2017 Jul 20];11(24):3273-83. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4976851/pdf/fon-11-3273.pdf
- Canadian cancer trials [Internet]. Toronto: Canadian Partnership Against Cancer Corporation. 2017 [cited 2017 Oct 18]. Available from: http://www.canadiancancertrials.ca/
- Triphase Research and Development I Corporation. Study of marizomib with temozolomide and radiotherapy in patients with newly diagnosed brain cancer. 2016 Sep 7 [cited 2017 Nov 8; updated 2017 Sep 25]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine; 2000 - . Available from: https://www.clinicaltrials.gov/ct2/show/NCT02903069 NLM Identifier: NCT02903069.
- Elsamadicy AA, Chongsathidkiet P, Desai R, Woroniecka K, Farber SH, Fecci PE, et al. Prospect of rindopepimut in the treatment of glioblastoma. Expert Opin Biol Ther. 2017 Apr;17(4):507-13.
- Northwest Biotherapeutics. Study of a drug [DCVax®-L] to treat newly diagnosed GBM brain cancer (GBM). 2002 Sep 17 [cited 2017 Sep 20; updated 2016 Oct 12]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine; 2000 - . Available from: https://www.clinicaltrials.gov/ct2/show/NCT00045968 NLM Identifier: NCT00045968.
- Sim HW, Morgan ER, Mason WP. Contemporary management of high-grade gliomas. CNS Oncol. 2017 Dec 15. Epub ahead of print.
- Sridhar P, Petrocca F. Regional delivery of chimeric antigen receptor (CAR) T-cells for cancer therapy. Cancers (Basel) [Internet]. 2017 Jul 18 [cited 2017 Sep 7];9(7). Available from: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC5532628
- O’Rourke DM, Nasrallah MP, Desai A, Melenhorst JJ, Mansfield K, Morrissette JJD, et al. A single dose of peripherally infused EGFRvIII-directed CAR T cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma. Sci Transl Med. 2017 Jul 19;9(399).
- Using ultrasound to deliver cancer therapies across the blood-brain barrier. Health Tech Update [Internet]. 2017 Jun [cited 2017 Sep 15];(18):9-10. Available from: https://www.cadth.ca/health-technology-update-issue-18
- Grisham J, Chesnut A, Dismuke D. The tumor: a non-legal thriller [Internet]. Charlottesville (VA): Focused Ultrasound Foundation; 2016. [cited 2017 Sep 26]. Available from: https://www.fusfoundation.org/download-the-tumor-by-john-grisham
- Nativis [Internet]. Seattle (WA): Nativis, Inc. 2017 [cited 2017 Sep 15]. Available from: http://nativis.com/
- Pfaff K. Nativis expands recurrent glioblastoma multiforme study to more patients . BioWorld MedTech. 2017 Sep 15;25(179):1,5.
- Green AL, Mulcahy Levy JM, Vibhakar R, Hemenway M, Madden J, Foreman N, et al. Tumor treating fields in pediatric high-grade glioma. Childs Nerv Syst. 2017 May 3;33:1043-5.
- Pediatric Brain Tumor Consortium. Feasibility trial of Optune for children with recurrent or progressive supratentorial high-grade glioma and ependymoma. 2017 Jan 17 [cited 2017 Sep 20]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine; 2000 - . Available from: https://www.clinicaltrials.gov/ct2/show/NCT03033992 NLM Identifier: NCT03033992.
- Chaudhry A, Benson L, Varshaver M, Farber O, Weinberg U, Kirson E, et al. NovoTTFTM-100A system (tumor treating fields) transducer array layout planning for glioblastoma: a NovoTALTM system user study. World J Surg Oncol [Internet]. 2015 Nov 11 [cited 2017 Jul 20];13:316. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4642621/pdf/12957_2015_Article_722.pdf
- Taillibert S, Le Rhun E, Chamberlain MC. Tumor treating fields: a new standard treatment for glioblastoma? Curr Opin Neurol. 2015 Dec;28(6):659-64.
- Zhang I, Knisely JP. Tumor-treating fields-a fundamental change in locoregional management for glioblastoma. JAMA Oncol. 2016 Jun 1;2(6):813-4.
- Weiss J. What I don’t have, what I can’t have: an obvious cure for brain cancer. Dallas Morning News [Internet]. 2017 Jun 13 [cited 2017 Sep 26]. Available from: https://www.dallasnews.com/life/healthy-living/2017/06/13/obvious-cure-brain-cancer
- Weiss J. Column: my way to the egress [Internet]. Washington (DC): Religion News Service; 2017. [cited 2017 Sep 26]. Available from: http://religionnews.com/columns/jeffrey-weiss/
- Marcione M. A ‘sci-fi’ cancer therapy fights brain tumors, study finds [Internet]. Boston (MA): STAT; 2017 Apr 2. [cited 2017 Oct 31]. Available from: https://www.statnews.com/2017/04/02/cancer-therapy-brain-tumors/
- Pandey M, Lands L, Baughman B, Michael M, Sorenson J, Weir A, et al. Patterns of compliance in the use of tumor treating fields (Optune) for brain tumors. Neuro Oncol [Internet]. 2016 [cited 2017 Jul 20];18(suppl 6):vi8. Available from: https://academic.oup.com/neuro-oncology/article-abstract/18/suppl_6/vi8/2541847/ (Presented at The 21st Annual Scientific Meeting and Education Day of the Society for Neuro-Oncology; 2016 Nov 17-20; Scottsdale, AZ).
- Lok E, San P, Hua V, Phung M, Wong ET. Analysis of physical characteristics of tumor treating fields for human glioblastoma. Cancer Med [Internet]. 2017 May 23 [cited 2017 Jul 20];6(6):1286-300. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5463092/pdf/CAM4-6-1286.pdf
- Harris D, Kumar V. Enhanced therapeutic benefits of tumor treating fields (TTfields) on superficially located glioblastoma multiforme (GBM). Neuro Oncol [Internet]. 2016 [cited 2017 Jul 20];18(Suppl 6):vi180. Available from: https://academic.oup.com/neuro-oncology/article-abstract/18/suppl_6/vi180/2543283/ (Presented at The 21st Annual Scientific Meeting and Education Day of the Society for Neuro-Oncology; 2016 Nov 17-20; Scottsdale, AZ).
- Mohan S, Chawla S, Skolnik A, Poptani H. Perspective on the EF-14 trial and its implications for the role of tumor-treating fields in the management of glioblastoma. Transl Cancer Res. 2016;5(Suppl 2):S272-S275.
- Batchelor T, Shih HA, Carter BS. Management of recurrent high-grade gilomas. In: Post TW, editor. UpToDate [Internet]. Waltham (MA): UpToDate; 2017 May 2 [cited 2017 Aug 29]. Available from: www.uptodate.com Subscription required.
- Tumour treating fields for glioblastoma multiforme [Internet]. Brisbane (AU): HealthPACT; 2012 May. [cited 2017 Sep 22]. Available from: https://www.health.qld.gov.au/__data/assets/pdf_file/0027/426816/wp068.pdf
- Press release, German Federal Joint Committee announces decision to support a clinical trial to study Optune® for the treatment of newly diagnosed glioblastoma [Internet]. St. Helier (Jersey Isle): Novocure; 2017 Sep 5. [cited 2017 Sep 26]. Available from: https://www.novocure.com/german-federal-joint-committee-announces-decision-to-support-a-clinical-trial-to-study-optune-for-the-treatment-of-newly-diagnosed-glioblastoma/
About This Document
Authors: Leigh-Ann Topfer, Kelly Farrah
Cite As: Alternating electric fields (“tumour-treating fields”) for the treatment of glioblastoma. Ottawa: CADTH; 2018 Jan. (CADTH issues in emerging health technologies; issue 165).
Acknowledgments: CADTH thanks the external reviewers who kindly provided comments on an earlier draft of this bulletin.
ISSN: 1488-6324 (online)
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policymakers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Contact [email protected] with inquiries about this notice or legal matters relating to CADTH services.
Dernière mise à jour : 16 janvier 2018

