Last Updated : October 29, 2010
Details
FilesProject Line:
Health Technology Review
Medication
In combination with lifestyle measures (weight control, proper nutrition, and adequate exercise), medications, such as insulin, play an important role in managing type 1, type 2, or gestational diabetes mellitus. Insulin is a human hormone that regulates the amount of glucose (sugar) found in the blood.
However, treatment with human insulin does not always replicate how our bodies normally secrete insulin, so this might not be the best way to control blood glucose levels. To address this limitation, insulin analogues were developed. Insulin analogues differ slightly in their chemical makeup from human insulins. Two main types of insulin analogues are available:
- Rapid-acting (or bolus) insulin analogues taken at mealtimes to control spikes in glucose
- Long-acting (or basal) insulin analogues taken to keep glucose levels steady in the background,
Need for Recommendations
Health care providers, consumers, and policy-makers require timely, evidence-based information they can rely on to help treat diabetes.
Because insulin analogues cost more than conventional human insulins, and health care dollars play a significant role in the sustainability of our health care system, the Canadian Optimal Medication Prescribing and Utilization Service (COMPUS) Advisory Committee (one of CADTH's former committees representing Canadian health jurisdictions) recommended that we review the clinical and economic evidence regarding the role of rapid-acting and long-acting insulin analogues in the treatment of diabetes mellitus.
Scope of CADTH Work on Insulin Analogue Therapy Topic
Key research questions on this topic area included:
- Are insulin analogues justified for all patients with diabetes?
- Do insulin analogues improve long-term health outcomes or patient quality of life?
CADTH conducted this project under its former COMPUS program. Insulin analogue therapy is one of a number of topics that CADTH has taken on in conjunction with our diabetes priority theme area.
Key Messages
CADTH's work on this project has resulted in the following key messages:
Bolus insulin therapy:
- In patients with type 1 diabetes, either regular human insulin or rapid-acting insulin analogues can be considered as first-line therapy (except in adolescent patients).
- In adolescent patients with type 1 diabetes, rapid-acting insulin analogues may be considered as first-line therapy.
- In patients with type 2 diabetes requiring bolus insulin, regular human insulin may be considered first. Although the evidence is limited and inconsistent, patients who are experiencing significant hypoglycemia while taking human insulin may benefit from rapid-acting insulin analogues.
Basal insulin therapy:
- In patients with type 1 or type 2 diabetes requiring basal insulin, insulin NPH should be considered first. Although the evidence is limited and inconsistent, patients who are experiencing significant hypoglycemia while taking insulin NPH may benefit from long-acting insulin analogues.
Tools
The Canadian Agency for Drugs and Technologies in Health (CADTH) offers tools and materials to assist with implementing its recommendations on optimal use of insulin analogues. Designed to support decision-making, the tools provide summaries of key information. To make these tools relevant to different audiences, CADTH can tailor specific tools to individual needs and settings. Contact us if you would like our assistance in tailoring any of these tools to meet your unique needs as a health care provider, policy-maker, or consumer.
To help decision-makers evaluate optimal use initiatives, CADTH has also developed a series of generic tools that can be applied across health topics. These generic tools including an audit and feedback guide, an evaluation framework, and academic detailing templates.
Insulin Analogue Therapy Tools
- Using Evidence in the Real World — a didactic presentation that provides a basic overview of evidence-based decision-making, using CADTH's work on self-monitoring of blood glucose and insulin analogues as examples.
- Guide to Starting and Adjusting Insulin for Type 2 Diabetes — a fold-out information card providing health care professionals with guidance on how and when to start insulin.
- Didactic Presentation for Pharmacists — designed for pharmacist education meetings, these slides may be used by continuing medical education providers, in part or in whole, for education on the optimal prescribing and use of insulin analogues.
- Didactic Presentation for Physicians — continuing medical education providers may use these presentation slides, in part or in whole, for education on the optimal prescribing and use of insulin analogues.
- Presentation for Pharmacists, together with Interactive Case Studies — these products are designed for use, in part or in whole, by continuing medical education providers in small education sessions regarding the optimal prescribing and use of insulin analogues for pharmacists.
- Presentation for Physicians, with Case Studies and a Participant's Guide — designed for continuing medical education providers for small education sessions on the optimal prescribing and use of insulin analogues; includes case studies.
- Project Highlights Brochure — highlights of the clinical and economic evidence from the CADTH insulin analogue project.
- Optimal Therapy Newsletter — summary of key clinical messages on optimal prescribing and use of insulin analogues for the treatment of diabetes to support decision-making by health care professionals.
- Quick Reference Prescribing Aid — contains comparative cost information and key messages about insulin analogues.
- COMPUS/RxFiles Pearls Document — detailed evidence and messaging “PEARLS,” which are key pieces of information around a topic area.
- Upskilling document — provides relevant reference material to qualified interventionists (i.e., continuing medical education providers, academic detailers).
- Interventions for Optimizing Therapy in Patients with Diabetes Mellitus: A Literature Review — a formal report summarizing study results on intervention strategies in diabetes therapy.
Published Articles
- Efficacy and safety of insulin analogues for the management of diabetes mellitus: a meta-analysis (Canadian Medical Association Journal)
- Cost-effectiveness of insulin analogues for diabetes mellitus (Canadian Medical Association Journal)
- Insulin analogues: too much noise about small benefits (Commentary; Canadian Medical Association Journal)
Related Information
- CADTH Webinar: Insulin Preparations – Which Types for Which Patients?
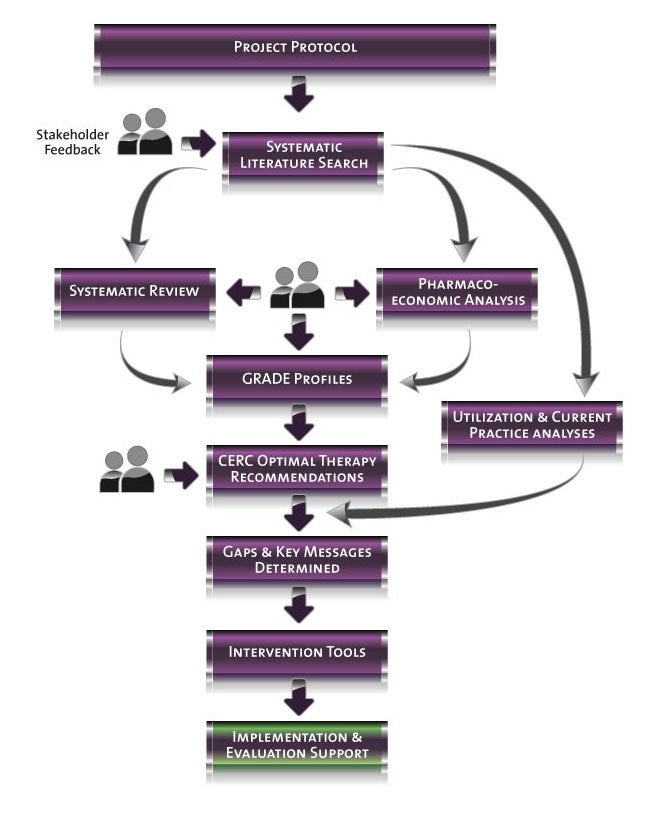
Project Status
 Represents current activity in
Represents current activity in
project process.
Reports
To assist health care providers, policy-makers, and consumers in making well-informed decisions about the prescribing and use of insulin analogues, our completed research and resulting recommendations are available in a series of Optimal Therapy Reports that provide:
- evidence-based clinical statements and cost-effectiveness statements
- a clear picture of current practice in Canada regarding the use of insulin analogues
- an analysis of the quantity of insulin agents prescribed, including market share and cost information
- an outline of gaps between current prescribing and use.
Summary Reports
- Project in Brief (one-page synopsis of clinical reports)
- Executive Summary (eight-page synopsis of clinical reports)
Final Reports
- Canadian Agency for Drugs and Technologies in Health. Optimal Therapy Recommendations for the Prescribing and Use of Insulin Analogues. Optimal Therapy Report – COMPUS 2009;2(7).
- Canadian Agency for Drugs and Technologies in Health. Gap Analysis and Key Messages for the Prescribing and Use of Insulin Analogues. Optimal Therapy Report – COMPUS 2008;2(8).
- Long-Acting Insulin Analogues for the Treatment of Diabetes Mellitus: Meta-analyses of Clinical Outcomes — Update of CADTH Technology Report No. 92. Optimal Therapy Report – COMPUS 2008;2(1).
- Summary of Revisions
- Canadian Agency for Drugs and Technologies in Health. Rapid-Acting Insulin Analogues for the Treatment of Diabetes Mellitus: Meta-analyses of Clinical Outcomes. Update of CADTH Technology Report No. 87. Optimal Therapy Report – COMPUS 2008;2(2).
- Canadian Agency for Drugs and Technologies in Health. GRADE Evidence Profiles on Long- and Rapid-Acting Insulin Analogues for the Treatment of Diabetes Mellitus. Optimal Therapy Report – COMPUS 2008;2(3).
- The Canadian Agency for Drugs and Technologies in Health. An Economic Evaluation of Insulin Analogues for the Treatment of Patients with Type 1 and Type 2 Diabetes Mellitus in Canada. Optimal Therapy Report − COMPUS 2008;2(4).
- The Canadian Agency for Drugs and Technologies in Health. Current Utilization of Insulin Products in Canada. Optimal Therapy Report − COMPUS 2008;2(5).
- Canadian Agency for Drugs and Technologies in Health. Current Practice Analysis: Insulin Analogues. A Qualitative Analysis of Canadian Physician Perceptions and Use of Insulin Analogues. Optimal Therapy Report – COMPUS 2008;2(6).
Files
Last Updated : October 29, 2010